Virtual Clinical Trials Market Growth Opportunities and Forecast till 2030
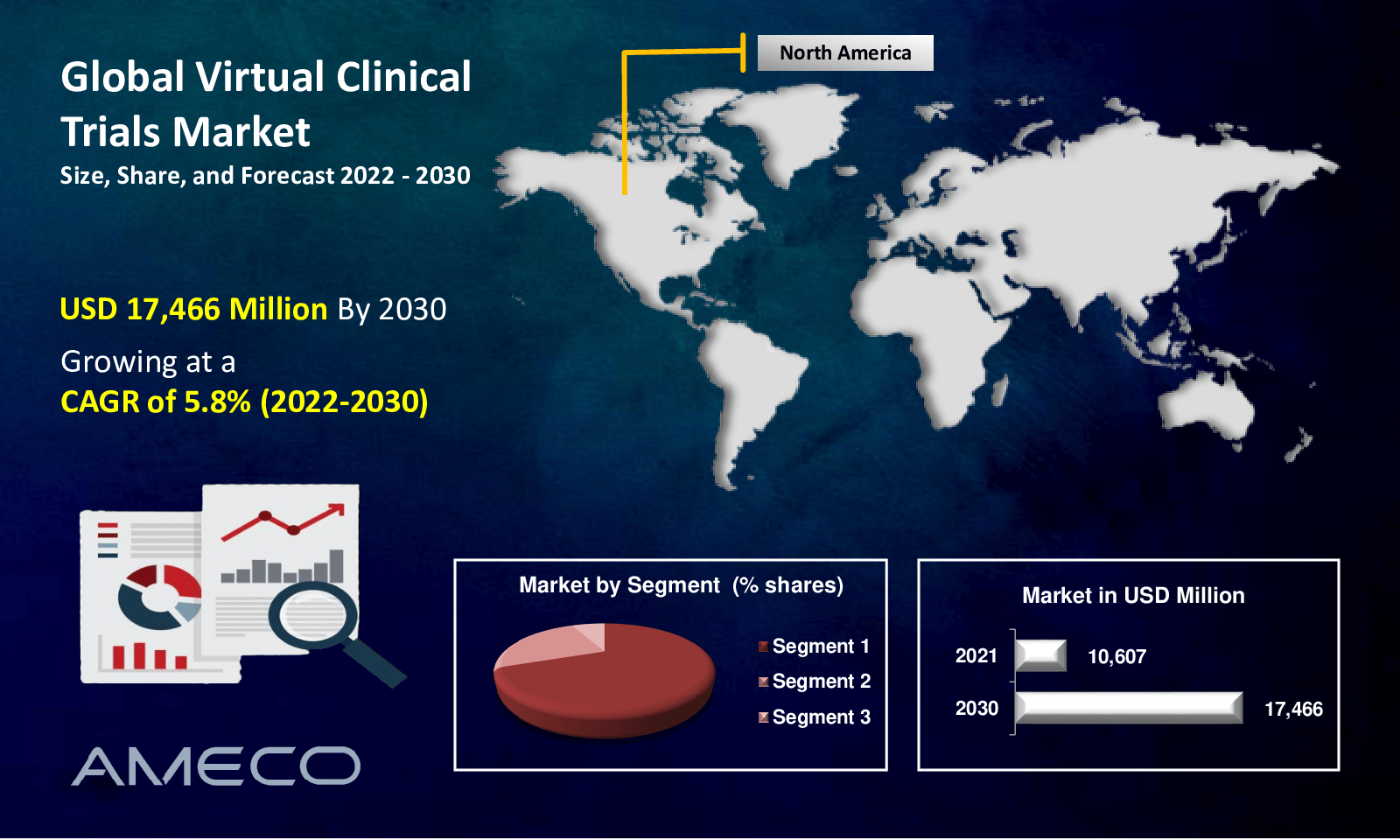
The global Virtual Clinical Trials Market size accounted for US$ 10,607 Million in 2021 and is projected to reach US$ 17,466 Million by 2030 with a CAGR of 5.8% from 2022 to 2030.
Virtual Clinical Trials (VCTs), also known as remote or decentralized trials, are a recent and underutilized approach to conducting clinical research that makes the most of modern technologies like apps, online social engagement platforms, and electronic monitoring devices. It can take up to 15 years to complete the labor-intensive and complex process of developing a clinical drug. An estimated US$ 2.6 billion is needed to develop a new drug from research and innovation to marketing approval. Only half of therapies that make it to phase 3 are approved, and about 85% of therapies fail during early clinical development. The clinical-testing phase accounts for more than two-thirds of the discovery and development of a new drug's overall cost, both financially and in terms of time.
|
Parameter |
Virtual Clinical Trials Market |
|
Virtual Clinical Trials Market Size in 2021 |
US$ 10,607 Million |
|
Virtual Clinical Trials Market Forecast By 2030 |
US$ 17,466 Million |
|
Virtual Clinical Trials Market CAGR During 2022 – 2030 |
5.8% |
|
Virtual Clinical Trials Market Analysis Period |
2018 - 2030 |
|
Virtual Clinical Trials Market Base Year |
2021 |
|
Virtual Clinical Trials Market Forecast Data |
2022 - 2030 |
|
Segments Covered |
By Study Design, By Indication, and By Region |
|
Virtual Clinical Trials Market Regional Scope |
North America, Europe, Asia Pacific, Latin America, and Middle East & Africa |
|
Key Companies Profiled |
IQVIA, Covance Inc., ICON plc, Parexel International, Medable, Inc. PRA Health Sciences, Oracle Corporation, LEO Innovation Lab, CRF Health, Medidata Solutions, Clinical Ink, Inc. |
|
Report Coverage |
Market Trends, Drivers, Restraints, Competitive Analysis, Player Profiling, Regulation Analysis |
Market Dynamics
To improve the patient-centric ecosystem, virtual clinical trials have the capacity to promote significant digital changes in clinical research methodologies. These solutions take into account the countless advantages of technology, cutting-edge applications, gadgets, online social networking sites, robotic process automation, machine learning, and others. Virtual clinical trials unquestionably make use of tele-health/digital technology to conduct secure and superior clinical trial research by incorporating virtual patient monitoring, wearable medical devices, remote SDV, etc. These virtual trials are convenient, affordable, and patient-focused.
Virtual clinical trials may be able to overcome the difficulties associated with traditional clinical trials, where finding suitable participants requires a lot of time and money because of their limited reach. Traditional and digital patient recruitment methods are both used in VCTs. The combination of recruitment strategies helps to more quickly hit the enrollment goal, which prompts a quicker start to the study. Patients are directly targeted when it happens digitally using web-based platforms, like search engines and social media. This strategy enables quick patient recruitment without regard to location, reaching potential trial participants everywhere.
Covid-19 Impact on Global Virtual Clinical Trials Market
Due to COVID-19 lockdowns and social distancing measures, a sizable number of companies announced delays to upcoming and ongoing clinical trials. Nearly 1,000 companies were impacted by a peak of 1,265 interrupted trials in June 2020. Trials that were already underway or that were scheduled to begin later had their enrollment halted or were completely abandoned. Since June 2020, there have been fewer disrupted trials; the majority of these disruptions are currently attributable to slow enrollment.
Market Insights
Study design, indication, and region are the three categories of the global virtual clinical trials industry. Based on the study design, the segmentation includes observational, expanded access, and interventional. According to our research, the interventional segment will dominate the virtual clinical trials market in the coming years.
The indication segment is further categorized into cardiovascular, oncology, and others. Among them, oncology segment achieved a significant amount of market share in 2021 and is expected to continue its trend throughout the forthcoming years.
Global Virtual Clinical Trials Market Geographical Competition
The global virtual clinical trials industry is divided into North America, Europe, Latin America, Asia-Pacific, and the Middle East and Africa. Historically, North America has dominated the market for virtual clinical trials, and this trend is anticipated to continue during the forecast period. The area is anticipated to continue to rule during the forecast period. This is due to increased R&D in this area, which is one of the key elements fueling the expansion of the global market for virtual clinical trials. On the other hand, the CAGR for Asia Pacific is anticipated to grow at the fastest rate over the next few years. This is because there is a larger patient pool available more often, making it simpler to find candidates and allowing for a greater uptake of digital technologies in the area. Additionally, the Coronavirus outbreak is anticipated to increase telemedicine adoption, spurring market growth.
Global Virtual Clinical Trials Industry Segment Analysis
Virtual Clinical Trials Market By Study Design
· Interventional
· Expanded Access
· Observational
Virtual Clinical Trials Market By Indication
· Oncology
· Cardiovascular
· Others
Virtual Clinical Trials Market Leading Companies
This section of the study honors the market's top vendors IQVIA, Covance Inc., ICON plc, Parexel International, Medable, Inc. PRA Health Sciences, Oracle Corporation, LEO Innovation Lab, CRF Health, Medidata Solutions, Clinical Ink, Inc., are the key companies mentioned in the research.
Virtual Clinical Trials Market Regions
North America
· U.S.
· Canada
Europe
· U.K.
· Germany
· France
· Spain
· Rest of Europe
Latin America
· Brazil
· Mexico
· Rest of Latin America
Asia-Pacific
· China
· Japan
· India
· Australia
· South Korea
· Rest of Asia-Pacific
Middle East & Africa
· GCC
· South Africa
· Rest of Middle East & Africa